Ultrafast reaction dynamics of the complete photo cycle of an indolylfulgimide studied by absorption, fluorescence and vibrational spectroscopy
13-Feb-2008
Journal of Molecular Liquids, 2008, 141, 130-6 published on 13.02.2008
Journal of Molecular Liquids, online article
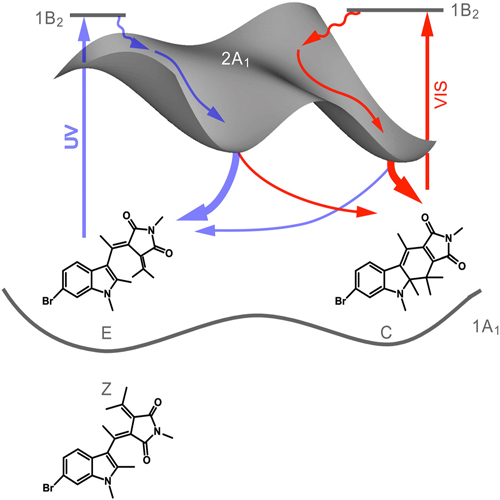
The reaction dynamics of the complete photo cycle, ring-opening (C→E) and ring-closure (E→C), of N-methyl-(6-bromo-1,2-dimethyl-3-indolyl) fulgimide dissolved in acetonitrile are analysed via steady-state and ultrafast spectroscopy using transient absorption in the UV/VIS and mid-IR complemented by ultrafast fluorescence broad-band spectroscopy with a Kerr-gate setup. For the C → E ring-opening reaction induced by light at ~550 nm, a time constant of ~3 ps was found for the S1 decay and the S0 repopulation. Non-exponential signatures, which occur in the 10 ps time domain, were observed and are assigned to the cooling of hot molecules in the electronic ground state. The E→C reaction dynamics induced by UVlight pulses at 360 nmand 387 nmoccurwithin less than 1 ps and are followed by vibrational cooling on the 10 ps time domain. Thus, both ring-opening and ring-closure reactions are completed within a few picoseconds. From transient IR studies and fluorescence measurements it is concluded that these pericyclic reactions occur from different geometries on the excited state potential energy surface.